Background: Exagamglogene autotemcel (exa-cel) is a non-viral cell therapy designed to reactivate fetal hemoglobin (HbF) via ex vivo CRISPR-Cas9 gene-editing of autologous CD34+ hematopoietic stem and progenitor cells (HSPCs) at the erythroid-specific enhancer region of BCL11A in patients (pts) with transfusion-dependent β-thalassemia (TDT). Here we report that in a pre-specified interim analysis, the pivotal CLIMB THAL-111 trial of exa-cel met primary and key secondary endpoints.
Methods: CLIMB THAL-111 is an ongoing, 24-month (mo), phase 3 trial of exa-cel in pts age 12-35y with TDT and a history of ≥100 mL/kg/y or ≥10 U/y packed red blood cell (RBC) transfusions in the 2y before screening. Primary endpoint is transfusion independence defined as proportion of pts maintaining a weighted average hemoglobin (Hb) ≥9 g/dL without RBC transfusion for ≥12 consecutive mos (TI12). Key secondary endpoint is proportion of pts maintaining a weighted average Hb ≥9 g/dL without RBC transfusion for ≥6 consecutive mos (TI6). Evaluable pts had ≥16 mos of follow-up after exa-cel infusion. Evaluation of TI12 and TI6 started 60 days after last RBC transfusion for post-transplant support or TDT management. Pts completing trial enrolled in long-term follow-up Study 131. Mean (SD) shown except where noted.
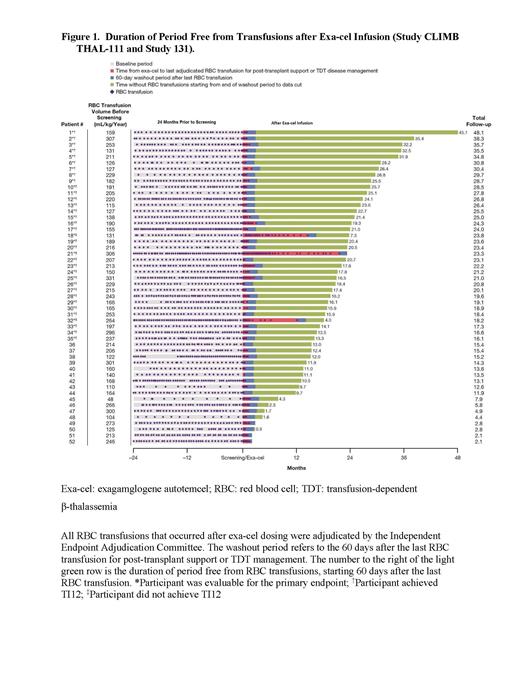
Results: As of 16 Jan 2023, 52 pts (mean age 21.5[range 12-35]y; 18[34.6%] age ≥12 to <18y; 31[59.6%] with severe genotypes [β 0/β 0 or β 0/β 0-like], median annualized transfusion volume 201.0 mL/kg) received exa-cel; median follow-up 20.4 (range 2.1-48.1) mos. Following infusion, all pts engrafted neutrophils and platelets (median 29 and 44 days, respectively). Of the 35 pts evaluable for primary and key secondary endpoints, 32 (91.4%) achieved TI12 and TI6 (95% CI: 76.9%, 98.2%; P<0.0001). Pts achieving TI12 stopped transfusions 35.2 (SD, 18.5) days after exa-cel infusion and remained transfusion independent for 22.5 (range, 13.3, 45.1) mos (Fig). For 3 pts not achieving TI12, one had reductions in annualized RBC transfusion volume of 83.9%, while the others have been transfusion-free for 7.3 mos and 4.0 mos starting 60 days after the last transfusion. For all pts, total Hb was 11.4 g/dL at Month 3 (≥12g/dL Month 6 onward) and HbF was 7.7 g/dL at Month 3 (≥ 10 g/dL Month 6 onward) with pancellular distribution (≥95% RBCs expressing HbF Month 6 onward). Proportion of edited BCL11A alleles was stable over time in bone marrow CD34 + and peripheral blood nucleated cells. Pts not yet evaluable and with sufficient follow-up were also transfusion-free. Quality-of-life (QOL) measures showed clinically significant improvements.
All pts had ≥1 adverse event (AE), most were Grade 1 or 2; 46 (88.5%) pts had AEs of Grade 3 or 4 severity. Most common AEs were febrile neutropenia (61.5%), headache (53.8%), and stomatitis (50.0%). Most AEs and serious AEs (SAEs) occurred within first 6 mos after infusion. Two pts had SAEs considered related to exa-cel: headache, hemophagocytic lymphohistiocytosis (HLH), acute respiratory distress syndrome and idiopathic pneumonia syndrome (latter also considered related to busulfan) all in the context of HLH (n=1) and delayed engraftment and thrombocytopenia (both also considered related to busulfan) (n=1), which all resolved. There were no deaths, discontinuations, or malignancies.
Conclusions: The CLIMB THAL-111 trial met primary and key secondary endpoints, with exa-cel treatment resulting in early and sustained increases in Hb and HbF leading to transfusion independence in >90% of pts with TDT and improved QOL. Safety profile of exa-cel was generally consistent with myeloablative busulfan conditioning and autologous transplantation. These results show exa-cel has the potential to deliver a one-time functional cure to pts with TDT.
Disclosures
Meisel:Gilead/KITE: Research Funding; CELGENE BMS: Consultancy, Research Funding, Speakers Bureau; CRISPR Therapeutics: Consultancy, Research Funding, Speakers Bureau; Vertex: Consultancy, Research Funding, Speakers Bureau; Novartis: Consultancy, Research Funding, Speakers Bureau; Miltenyi Biotech: Research Funding; Bluebird Bio: Consultancy, Speakers Bureau; medac: Consultancy, Research Funding, Speakers Bureau. Shah:Vertex: Membership on an entity's Board of Directors or advisory committees. Carpenter:Vertex Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees; BlueBird Bio: Membership on an entity's Board of Directors or advisory committees. Kwiatkowski:Regeneron Pharmaceuticals: Consultancy; Bristol Myers Squibb: Consultancy; BioMarin Pharmaceutical: Consultancy; Vertex Pharmaceuticals: Consultancy; Forma Therapeutics: Consultancy, Research Funding; Chiesi Farmaceutici: Consultancy; Bluebird Bio: Research Funding; Agios Pharmaceuticals: Consultancy, Research Funding; Editas Medicine: Research Funding; Pfizer: Research Funding. Mapara:Crispr/vertex: Consultancy; Incyte: Consultancy; Bluebird bio: Consultancy. Liem:Bluebird Bio: Research Funding; Vertex: Research Funding; NIH/NCATS: Research Funding; NIH/NHLBI: Research Funding; Editas: Research Funding; Global Blood Therapeutics: Research Funding. Algeri:Vertex Pharmaceuticals: Consultancy, Membership on an entity's Board of Directors or advisory committees. Kattamis:Agios Pharmaceuticals: Consultancy; Bristol Myers Squib/Celegene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Vertex Pharmaceuticals: Consultancy, Membership on an entity's Board of Directors or advisory committees; Chiesi: Honoraria; Novartis: Consultancy, Honoraria, Research Funding; Amgen: Consultancy; Ionis Pharmaceuticals: Consultancy; Vifor: Consultancy. Sheth:Agios: Consultancy, Other: Travel support, Research Funding; Bristol Myers Squibb/ Celegene: Consultancy, Other: Travel support, Research Funding; Bluebird bio: Consultancy, Other: Travel support; Fulcrum: Consultancy; Chiesi: Consultancy; CRISPR: Membership on an entity's Board of Directors or advisory committees; Vertex Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees. Grupp:Novartis: Consultancy, Research Funding; Vertex: Consultancy, Research Funding; Jazz: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Kite: Research Funding; Servier: Research Funding; CBMG: Consultancy, Membership on an entity's Board of Directors or advisory committees; Adaptimmune: Consultancy, Membership on an entity's Board of Directors or advisory committees; Cellectis: Consultancy, Membership on an entity's Board of Directors or advisory committees; Juno: Consultancy, Membership on an entity's Board of Directors or advisory committees; Allogene: Consultancy, Membership on an entity's Board of Directors or advisory committees; Cabaletta: Consultancy, Membership on an entity's Board of Directors or advisory committees. Kohli:Vertex Pharmaceuticals: Current Employment. Shi:Vertex Pharmaceuticals: Current Employment. Ross:Vertex Pharmaceuticals: Current Employment. Bobruff:Vertex Pharmaceuticals: Current Employment; CRISPR Therapeutics, Inc: Ended employment in the past 24 months. Simard:Vertex Pharmaceuticals: Current Employment. Zhang:Vertex Pharmaceuticals: Current Employment. Morrow:CRISPR Therapeutics: Current Employment, Membership on an entity's Board of Directors or advisory committees; Vertex Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees. Hobbs:Vertex Pharmaceuticals: Current Employment. Frangoul:Vertex Pharmaceuticals: Consultancy, Membership on an entity's Board of Directors or advisory committees; Editas Medicine: Consultancy; Jazz Pharmaceuticals: Speakers Bureau; Rocket Pharmaceuticals: Consultancy, Other: Member of DSMB for a study.